Cretaceous metabolic pathways influence present-day global nutrient cycles
Kiel research team, searching for evolutionary origins, discovers that foraminifera in oxygen minimum zones may cooperate with bacterial symbionts in nitrate respiration
In the context of climate change, the phenomenon of oxygen-depleted areas in the ocean has become a focus of scientific attention in recent years. These areas, known as oxygen minimum zones (OMZ), are located in the Indian Ocean or off the Peruvian coast in the Pacific, for example. Depending on water depth, little or no oxygen is found there. In a new study, a transdisciplinary research team with the participation of Kiel University and GEOMAR Helmholtz Centre for Ocean Research Kiel was able to gather previously unknown details about the origin and adaptation of certain unicellular organisms to these special environmental conditions and their influence on the marine nutrient cycle: Because their metabolism does not rely on oxygen, numerous species of foraminifera are found in the OMZ. These are unicellular, shell-forming microorganisms that already feature a cell nucleus and thus belong to the so-called eukaryotes.
Their special lifestyle is based on so-called anaerobic respiration or nitrate respiration, in which nitrate available in the water is converted into molecular nitrogen in the absence of oxygen. Nitrogen, as a basic nutrient of all living organisms, is no longer biologically available as a result of this process and is thus lost to the marine habitat. The research team led by Professor Tal Dagan of the Institute of General Microbiology at Kiel University has now been able to prove via genome analyses that the ability of nitrate respiration, still preserved in most foraminifera today, might have originated in a common predecessor around 100 million years ago. Their analyses also showed that the foraminifera probably rely on bacterial support for a specific step in the process of nitrate respiration, also known as denitrification, and are therefore associated with certain symbiotic bacterial species. The Kiel researchers, who are active in the Kiel Evolution Center (KEC) and the Collaborative Research Center (CRC) 1182 “Origin and Function of Metaorganisms”, published their results together with international colleagues recently in the renowned journal Proceedings of the National Academy of Sciences of the United States of America (PNAS).
Nitrate respiration originated millions of years ago
The research team examined sediment samples from the Peruvian OMZ and was able to identify around ten different species of foraminifera living in the seafloor. From previous research almost all of these species are known to be capable of nitrate respiration. The scientists therefore looked for genes that are likely involved in nitrate metabolism. “Indeed, numerous of these genes were found in our samples from Peru. Since these species have been shown to perform nitrate respiration, we were thus able to provide a causal link between the presence of the genes and their function,” emphasizes first author Dr Christian Wöhle, a former member of Dagan’s Genomic Microbiology group at Kiel University.
In a next step, the researchers traced back how long ago the evolutionary acquisition of nitrate respiration could have occurred. To do this, they created so-called phylogenetic trees, which visualize the relationships between different species to identify a possible common ancestor. “In this way, we were able to narrow down a time period for three specific groups of foraminifera. All earlier species lacked the genetic equipment for nitrate respiration,” Wöhle explains. “Thus, we could reconstruct that they probably go back to a common predecessor that must have developed this trait in the Cretaceous period about 100 million years ago,” Wöhle continues.
Microbes provide missing metabolic step
In a metagenome analysis of the Peruvian sample material, the researchers also examined the genetic material of all species found to establish possible symbiotic relationships between various foraminifera and bacteria. First, comparisons with foraminifera species from distant locations showed that they are often associated with a similar and stable composition of bacterial species, regardless of geography. “In our study, we further noticed an association between a particular genus of foraminifera and so-called Desulfobacteria. These bacteria have previously been observed to be colonizers of other groups of foraminifera,” said Dr Alexandra-Sophie Roy, a former member in the Genomic Microbiology group.
Another indication of a symbiotic relationship between these bacteria and the denitrifying foraminifera was found in the form of the so-called NapA gene. In Desulfobacteria, it is responsible for an enzyme that can initiate the start of the multistep process of nitrate respiration. “Earlier studies showed that precisely this first step cannot be performed by foraminifera themselves. Therefore, it seems plausible that the bacteria take over this part of nitrate respiration, for example by converting nitrate into nitrite that is useful for their hosts, and therefore form a stable symbiosis with the foraminifera,” Roy explains.
New insights into global nutrient cycles
“Our work on the origins and evolution of the genetic basis of ancient nitrate metabolism provides us with important new insights to better understand today’s global biogeochemical cycles,” summarizes Professor Tal Dagan, KEC board member and project leader in the CRC 1182. In particular, she said, the possible involvement of bacteria in nitrate respiration in the oxygen-depleted ocean is an important but so far understudied component that needs further research in the future. “The accelerating pace of global environmental change requires deepened knowledge of the turnover and distribution of elemental substances in the ocean. Our new results contribute to a better understanding of the impact of climate change on oxygen-dependent nutrient cycles in the ocean and their effects on marine organisms,” adds Dr Joachim Schönfeld from GEOMAR, who was also involved in the study.
Scientific contact:
Prof. Tal Dagan
Genomic Microbiology, Head
Institute for General Microbiology, Kiel University
+49 (0) 431 880-5712
tdagan@ifam.uni-kiel.de
More information:
- Genomic Microbiology (Dagan group),
Institute for General Microbiology, Kiel University:
www.mikrobio.uni-kiel.de/de/ag-dagan
- Kiel Evolution Center (KEC), Kiel University:
www.kec.uni-kiel.de
- Collaborative Research Center 1182
“Origin and Function of Metaorganisms“, Kiel University:
www.metaorganism-research.com
Original publication:
Christian Woehle, Alexandra-Sophie Roy, Nicolaas Glock, Jan Michels, Tanita Wein, Julia Weissenbach, Dennis Romero, Claas Hiebenthal, Stanislav N. Gorb, Joachim Schönfeld, Tal Dagan (2022): Denitrification in foraminifera has an ancient origin and is complemented by associated bacteria. PNAS First published on 15. June 2022.
DOI: 10.1073/pnas.2200198119

© Jan Michels

Dr Christian Woehle (left) and Dr Joachim Schönfeld handling a multicorer during a sampling expedition in Kristineberg, Sweden 2015.
© Prof. Tal Dagan

© Prof. Tal Dagan

The researchers obtained the foraminifera investigated from such polycarbonate core tubes taken from the Pacific seabed at a depth of approximately 500 meters.
© Prof. Tal Dagan
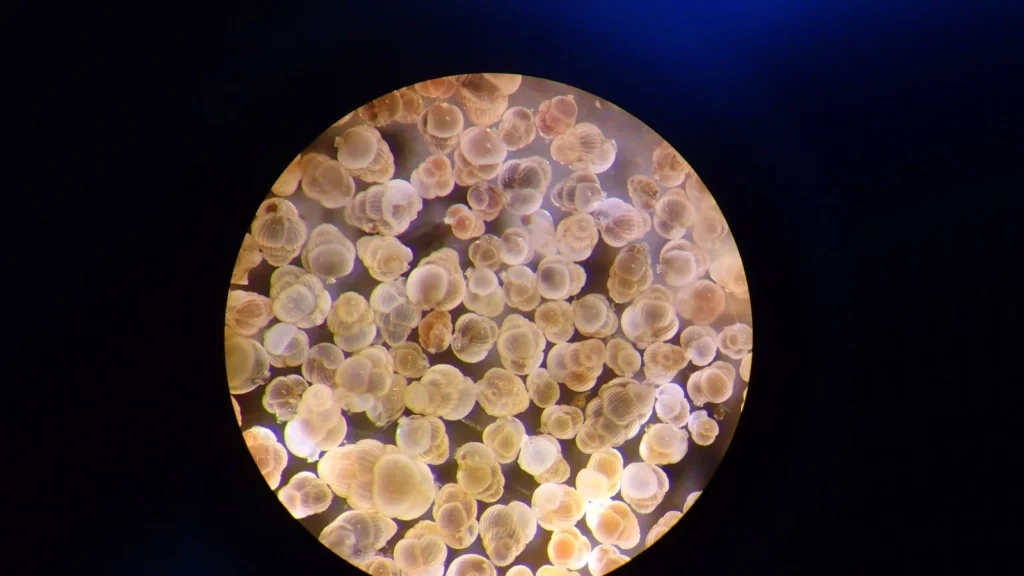
Some specimens of the studied species Uvigerina striata sieved out of the sediment using sieves of various sizes.
© Prof. Tal Dagan





