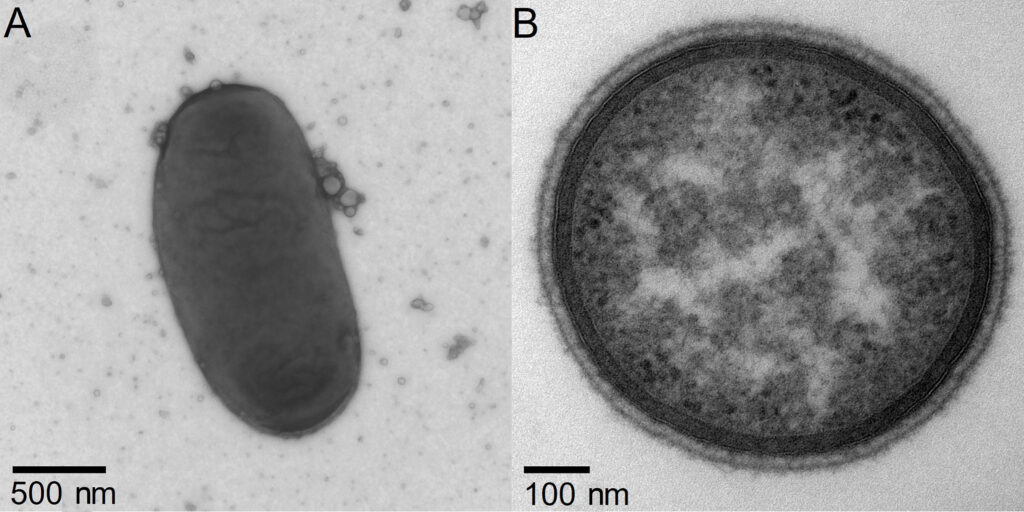
Similar phage communities in human and primate intestines
International research team with Kiel participation explores ecological and evolutionary origins of phage communities as part of the human microbiome
We live in a world dominated by microorganisms. They existed on earth long before the first multicellular organisms came into being. As part of a metaorganism, i.e. the community of a complex living being with colonizing microorganisms, they remain a central component of life on our planet today and are of great importance for the health of humans, animals and plants. This microbial colonization of a living being, known as the microbiome, also includes various viruses. In the case of viruses associated with humans, these are mostly phages found in the intestine, which are viruses that infect bacteria. An international research team, with the participation of Kiel University, investigated the origins of phage communities in the human intestine. To this end, the Kiel scientists, together with researchers from the Robert Koch Institute (RKI), the Max Planck Institutes for Evolutionary Biology and Evolutionary Anthropology, North Carolina’s Duke University, and other partner institutions, analyzed the phages of non-human primates, the closest living relatives of humans. In a comparative approach, they examined the gut phages of 23 wild primate species found in widely varying ecosystems around the world. They compared these phage communities with those of humans living in Europe and Africa and found that closely related species of most phages associated with humans are detected in wild primates. At Kiel University, researchers within the Collaborative Research Center (CRC) 1182 “Origin and Function of Metaorganisms” contributed to this work through genome sequencing and data analysis, with results published in the renowned journal Proceedings of the National Academy of Sciences of the United States of America (PNAS).
Mirror image of primate evolution
The researchers first examined the evolutionary relationships of primate phages with each other. They found that for some groups of phages, their relationships mirror the evolutionary history of the primates that harbor them, an observation previously made for the bacterial component of the gut microbiome. This pattern, known scientifically as co-divergence, suggests that some phages maintained an association with particular primate strains over millions of years. Surprisingly, this was possible even though in some cases these primates shared the same ecosystem with other primates with divergent phage communities, and in some cases even consumed them regularly. Dr. Sébastien Calvignac-Spencer of the RKI explains: “We sometimes observe patterns of co-divergence between primates and the viruses that infect them, demonstrating that some viruses maintain a close relationship with their hosts over evolutionary timescales. But phages infect bacteria, not primate cells, and we were rather surprised to find that some phages also showed such a pattern.”
“The project is part of a larger research collaboration with Dr Fabian Leendertz’s group at the Robert Koch-Institute, for which we have produced sequencing data in Kiel that we are now jointly investigating in a variety of ways,” says Dr Malte Rühlemann from the Institute of Clinical Molecular Biology at Kiel University. “The goal of our project in the CRC 1182 is to understand the mechanisms that determine why the human microbiome looks the way we observe it. The evolutionary view of phage communities in humans and non-human primates represents an important component of this and is unique in this form to date,” Rühlemann continues.
Phages form long-term associations with hosts
Given that some phages form long-term associations with specific primate species, the scientists wanted to understand how such long-term associations are maintained. For example, they found that neighboring social groups of baboons harbor unique phage communities. In these groups, those animals that form lasting friendships and spend lots of time grooming one another have very similar phage communities – regardless of their genetic relatedness. First author Dr Jan Gogarten of the RKI notes, “Our study underscores the importance of long-term observational studies in wild primates, such as in the Amboseli Baboon Research Project in Kenya. This approach can answer questions that are difficult to study in humans. For example, some of these baboon groups have been observed for decades. Therefore, the relationships and social structures in the animal groups are very well known there. In addition, scientists often collect fecal samples that allow long-term monitoring of microbiome composition. Such detailed resolution of data does not exist for human populations.” However, the results suggest that social interactions in humans may also have an impact on phage transmission – something to be clarified in future research.
In a next step, the researchers investigated how flexible or permanent the connections between primates and their phage communities are. In principle, wild primates have maintained their phage composition over millions of years. The research team thus analyzed phages living in primates in zoos. They found that primates in captivity lose the phages they normally harbor in the wild. Overall, the study provides new insights into the evolutionary and ecological origins of phages associated with humans and opens interesting perspectives for further research.
Coauthor Professor John Baines, who leads the Section of Evolutionary Medicine at Kiel University adds, “Given the drastic changes in our own microbial communities that have occurred during the process of hominization and the current plague of civilization diseases associated with the microbiome, the importance of this priceless information on the microbial communities of our closest living relatives cannot be overstated.”
Original publication:
Gogarten J., Rühlemann, M., Archie, E., Tung, J., Akoua-Koffi, C., Bang, C., Deschner, T., Muyembe-Tamfun, J.-J., Robbins, M. M., Schubert, G., Surbeck, M., Wittig, R. M., Zuberbühler, K., Baines, J. F., Franke, A., Leendertz, F. H., and Calvignac-Spencer, S. (2021): Primate phageomes are structured by superhost phylogeny and environment. PNAS. DOI: 10.1073/pnas.2013535118
Images are available for download:
www.uni-kiel.de/de/pressemitteilungen/2019/272-rausch-microbiome-baines.jpg
Caption: Professor John Baines and his team from the Kiel CRC 1182 hope to draw important conclusions about the composition and changes in the human microbiome by studying the phage communities of non-human primates.
© Christian Urban, Kiel University
www.uni-kiel.de/de/pressemitteilungen/2021/006-ruehlemann-naturegen-author.jpg
Caption: Dr Malte Rühlemann, scientist at the IKMB, contributed to the new research by analyzing data generated by genome sequencing at Kiel University.
© Private
www.uni-kiel.de/de/pressemitteilungen/2021/082-gogarten-pnas-baboon.jpg
Caption: The international research team studied the intestinal phages of 23 wild primate species, including baboons (pictured), and compared them to phage communities in humans.
© William Warby via Wikimedia Commons, license: CC-BY-2.0
Contact:
Dr Malte Rühlemann
Genetics and Bioinformatic,
Institute for Clinical Molecular Biology, Kiel University:
Phone: +49 (0) 431-500-15132
Email: m.ruehlemann@ikmb.uni-kiel.de
Prof. John Baines
Section of Evolutionary Medicine, Head
Institute for Experimental Medicine, Kiel University:
Phone: +49 (0) 431-500-30310
Email: j.baines@iem.uni-kiel.de
Prof. Andre Franke,
Head of Genetics and Bioinformatic Group,
Institute for Clinical Molecular Biology, Kiel University:
Phone: +49 (0) 431 – 500-15 110
Email: a.franke@ikmb.uni-kiel.de
More information:
Section of Evolutionary Medicine,
Max-Planck-Institute for Evolutionary Biology, Plön / Kiel University:
http://web.evolbio.mpg.de/evolgenomics/index.html
Genetics and Bioinformatic Group,
Institute for Clinical Molecular Biology, Kiel University:
www.ikmb.uni-kiel.de/research/genetics-bioinformatics
Epidemiology of Highly Pathogenic Microorganisms,
Robert Koch-Institute:
www.rki.de/DE/Content/Forsch/Projektgruppen/Projektgruppe_3/P3_node.html